For the fastest service possible and immediate testing, please order below or call 866-843-4545.
National Drug Screening has testing locations in all areas of the United States.
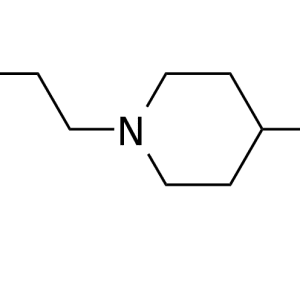
Order your drug test online. Immediate drug tests for individuals, DOT, non-DOT, company testing, courts, and more. Specimen types include urine, hair, Breath alcohol, and blood. EtG testing and 4-18 panel drug tests are available. Order online and you will receive the same business day instructions for your testing at the most convenient collection site location. Orders are processed Monday-Friday 9am-6pm ET. No account is necessary to order testing and once completed, results are sent to your e-mail. We specialize in providing fast and affordable drug testing services.