Oral fluid drug testing or sometimes called saliva drug testing is available throughout the United States. Because of the easy collection process, employers typically collect oral fluid specimens to send out to a laboratory or to perform an instant oral fluid drug test. DOT has approved oral fluid drug testing for transportation regulated employers and final rule takes effect June 1, 2023. See DOT Oral Fluid Drug Testing.
Oral fluid drug testing has gained popularity due in large part to it being perceived as less invasive and a perception that it is less expensive. The oral fluid drug test captures oral fluid with a device that appears to be a sponge on a stick. The donor inserts the oral fluid collection device into the mouth to collect the oral fluid. Most devices will take at least two minutes for the collection pad (sponge) to collect a sufficient amount of oral fluid. Oral fluid drug testing is detecting very recent use of drugs and often the parent drug as opposed to a metabolite.
Oral fluid drug testing is very suitable for workers in states that prohibit testing for marijuana metabolites, allowing only for the detection of the parent drug itself. Oral fluid drug testing is highly recommended for post-accident testing and reasonable suspicion testing because of the detection of the very recent use of illicit drugs.
Oral fluid drug testing topics to be covered on this page include:
- Lab Based Oral Fluid Drug Testing
- Lab Based Oral Fluid Panels Available
- Instant Oral Fluid Drug Testing
- Instant Oral Fluid Drug Testing Panels
- Proctored Oral Fluid Drug Testing
- DOT and Oral Fluid Drug Testing
- Oral Fluid vs Urine Drug Testing
- Oral Fluid Drug Testing And Potential Cost Savings
- Benefits of Oral Fluid Drug Test Self-collection

Lab Based Oral Fluid Drug Testing
With lab based oral fluid drug testing, an oral fluid or saliva specimen is collected and sent to the laboratory for screening and confirmation testing. The procedure at the lab is similar to urine drug testing. Oral fluid drug testing is sometimes referred to as saliva drug testing.
The oral fluid test is sometimes called a saliva test. The saliva specimen is collected with a mouth swab. The swab and specimen are shipped via FedEx to a certified laboratory for initial screening and confirmation testing when necessary.
The oral fluid drug test process detects drug use for up to 48 hours from time of collection. The drug test result comes back from the lab to the MRO. Positive rates are often about the same as urine drug screening although marijuana positive rates tend to be slightly higher.
Lab Based Oral Fluid Panels Available
Most labs use a collection device called Quantisal which has as a unique “volume adequacy indicator” that ensures enough specimen for screening, confirmation and repeat testing. Our labs offer panel options for oral fluid drug testing including:
- Five panel oral fluid drug test - amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, 6-AM), cocaine (metabolite), marijuana (THC), and phencyclidine (PCP).
- Five panel oral fluid drug test with alcohol – Ethyl alcohol, amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, 6-AM), cocaine (metabolite), marijuana (THC), and phencyclidine (PCP).
- Five panel oral fluid drug test with expanded opiates – amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, hydrocodone, hydromorphone, 6-AM), cocaine (metabolite), marijuana (THC), and phencyclidine (PCP).
- Four panel oral fluid drug test with expanded opiates – amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, hydrocodone, hydromorphone, 6-AM), cocaine (metabolite) and phencyclidine (PCP). Marijuana is not included on this panel.
- Nine panel oral fluid drug test with expanded opiates – amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, hydrocodone, hydromorphone, 6-AM), cocaine (metabolite), marijuana (THC), phencyclidine (PCP), barbiturates, Benzodiazepines and Propoxyphene
- Eight panel oral fluid drug test with expanded opiates – amphetamine, methamphetamines (including MDMA and its metabolite), opiates (codeine, morphine, hydrocodone, hydromorphone, 6-AM), cocaine (metabolite), phencyclidine (PCP), barbiturates, Benzodiazepines and Propoxyphene. Marijuana is not included on this panel.
Instant Oral Fluid Drug Testing
Oral fluid testing can also be accomplished with the use of a rapid or instant oral fluid device often called point of collection device. The biggest advantage is that negative results can be available in just a few minutes. This is a big advantage for drug testing for pre-employment, post-accident and reasonable suspicion. For all instant testing when the result is not negative, the device and specimen should be sealed up and sent to a laboratory for confirmation testing. Lab results then go to the MRO for review and verification. The use of a custody and control form (CCF) should be used to accompany the specimen to the lab.
Instant Oral Fluid Drug Testing Panels
National Drug Screening offers three in stock instant oral fluid devices that are listed below. Additional configurations can be customized for large volume orders.
- INSTANT ORAL FLUID 5 (NO THC) SELF COLLECT – Amphetamines, Methamphetamines, Cocaine, Opiates (Heroin, Morphine, Codeine) & PCP
- INSTANT ORAL FLUID 7 – Amphetamines, Methamphetamines, Cocaine, Opiates (Heroin, Morphine, Codeine), PCP, Oxycodone & Marijuana (THC)
- INSTANT ORAL FLUID 10 – Amphetamines, Methamphetamines, Cocaine, Opiates (Heroin, Morphine, Codeine), PCP, Oxycodone, Marijuana (THC), Barbiturates, Benzodiazepines, Methaqualone & Methadone

Proctored Oral Fluid Drug Testing
In remote areas or for folks working from home, new technology allow for self-collect proctored oral fluid drug testing. This is a very efficient testing solution favored by both employees and employers, providing rapid and precise results. Eliminate the need for uncomfortable urine samples and the lengthy wait of a week for results. Make immediate hiring decisions. Results are accessible in under 30 minutes from start to finish—without any of the associated inconveniences!
With proctored oral fluid drug testing the drug test kits are delivered to the employee or applicant to be tested. The collection process is supervised through a video conference. Any non-negative results are sent to the laboratory for verification while maintaining the chain of custody. Final lab results are verified and reported by a medical review officer (MRO).
5, 8 and 10 panel configurations are available for the proctored oral fluid drug testing available from National Screening. The 5 panel configuration does not contain testing for marijuana (THC).
- 10 PANEL: AMP, BAR, BZO, COC, MET, MTD, OPI, OXY, PCP, THC
- 8 PANEL: AMP, COC, MET, MTD, OPI, OXY, PCP, THC
- 5 PANEL: AMP, COC, MET, OPI, OXY
DOT and Oral Fluid Drug Testing
DOT now allows oral fluid drug testing for DOT regulated drug testing. A rule was proposed in the Mandatory Guidelines for Federal Workplace Drug Testing; it was opened for comments and the comment period closed on July 14, 2015, with 120 comments received. SAMHSA finalized the rule and added it to the Mandatory Guidelines for Federal Workplace Drug Testing; DOT has decided to adopt most of the provisions into 49 CFR Part 40 – the DOT rule for DOT drug and alcohol testing programs.
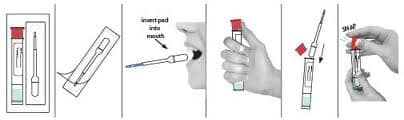
DOT has clearly adopted: split specimen collections and strict collector training including qualification and proficiency demonstration requirements. Laboratories and MRO’s will also have to come up to speed on the requirements that SAMHSA has written into the Mandatory Guidelines for Federal Workplace Drug Testing and DOT revisions to 49 CFR Part 40 that take effect 06/01/2023.
Oral fluid testing for DOT is always lab based testing and not instant oral fluid testing. Employers will have the only authority to choose whether an applicant or employee will be tested with urine or oral fluid. In some situations, such as shy bladder the employer cans switch from one specimen type to the other (example: urine to oral fluid). Learn more from National Drug Screening (NDS) about DOT Oral Fluid Drug Testing.
Oral Fluid vs Urine Drug Testing
Unlike urine drug screens, it is very difficult to cheat a saliva-based or oral fluid drug test as the collection is witnessed or directly observed. It is important however to carefully monitor the donor to ensure they do not introduce something onto the pad or collection vial. Labs are seeing a higher percentage of positive drug tests with oral fluid testing, particularly with marijuana. This may be due to the difficulty in cheating on an oral fluid drug test as opposed to urine drug testing and the detection window may also be a factor. Oral fluid drug testing will detect more recent use of the drug, particularly for marijuana.
One of the major differences between oral fluid drug testing and urine drug testing is the detection time. The detection time is different for each drug tested. The notable difference is that the detection time is shorter for oral fluid drug testing and recent drug use is more likely to be detected with oral fluid drug testing. The window of detection for oral fluid drug testing is from several hours after use to two days. The narrow but immediate detection window makes oral fluid drug testing a natural choice for reasonable suspicion testing and for post-accident testing.
Different labs are testing different drug panels utilizing oral fluid testing and we also see the labs using different collection devices. Below is a summary of what we have observed from the various labs in regard to oral fluid drug testing, the drugs being tested, and the collection devices being used.
Oral Fluid Drug Testing And Potential Cost Savings
It is often stated that oral fluid drug testing is less expensive than urine drug testing. That is typically only true when the employer is collecting the specimen, completing the custody and control form and processing the specimen out to the laboratory. The reason it is stated that oral fluid drug testing is less expensive is that the cost of the collector and the collection site is eliminated. The challenge for the employer is training a staff member to collect the oral fluid specimen, complete the custody and control form and process the specimen out to the laboratory. Each of these steps are absolutely essential to the oral fluid testing process and can have serious consequences if not completed properly.
Several years back, Publix Supermarkets, located in the southeast; replaced urine drug testing with oral fluid drug testing. This large retail grocery chain calculated hundreds of thousands of dollars in savings as a result of no longer utilizing collectors or collection sites. Store managers were trained to perform the oral fluid collections and processing out to the laboratory.
Implementing oral fluid testing in any business will require thought and action. Drug free workplace policies will need to be updated, and specimen collection procedures will need to be created including getting training for the employer staff to perform the specimen collections and process the specimens to the laboratory. Important decisions will have to be made that may dramatically affect the business. It is important to review carefully at all layers of management a decision to change from urine drug testing to oral fluid drug testing.

Benefits of Oral Fluid Drug Test Self-collection
Imagine again that you’re in that conference room we talked about earlier. What would your list of pros of self-collection look like? Maybe something like this:
- Complete control of collection process
- Less time away from work for employees
- Less opportunity for cheating
- Recent detection of drug used particularly important for post-accident drug testing and reasonable suspicion drug testing
- No more collection cost
Self-collection means the employer knows exactly when, where, and how collection takes place. There’s no need to coordinate testing hours of operation, hiring schedules and random events with a third party collection provider. This makes pre-employment testing easier and faster. It makes random testing truly random and a surprise for employees being tested. And in the event of an accident, it eliminates the need to send potentially injured employees to a collection site. Employees are away from work for an average of 15 minutes instead of two or three hours. All an employee has to do is walk to the manager’s office, swab, and go back to work. This approach also means that there is less opportunity for cheating. There’s no such thing as shy bladder when collecting oral fluid samples, nor can an employee swap out his oral fluid for his buddy’s. And of course, it costs less to do self-collection than a professional collection.
Conclusion
Oral fluid testing is an excellent way for employers to save money on their drug testing program while taking advantage of a lab-based test. When self-collection isn’t feasible in all instances, oral fluid testing can be combined with a different testing methodology or professional collectors can be used. As with any drug testing program, design and implementation are critical and each should be tailored to your organization’s program goals and corporate culture. And, as with any program, compliance with all federal and state drug testing laws, partnership with reputable TPA’s, manufacturers and laboratories, and an updated policy are critical to a successful program. DOT regulations for DOT oral fluid drug testing are critical to review, lean more at:

